|
When the pH of the solution equals pKa, the concentrations of HA and A(-) must be equal (log 1 = 0). Here, the pKa represents the acidity of a specific conjugate acid function (HA). In order to determine the nature of the molecular and ionic species that are present in aqueous solutions at different pH’s, we make use of the Henderson-Hasselbalch Equation, written below. Since amino acids, as well as peptides and proteins, incorporate both acidic and basic functional groups, the predominant molecular species present in an aqueous solution will depend on the pH of the solution. 6, reflecting the higher basicity of two-amines. For that 2-amino acidity proline, pKa2 is 10. 
4), and yet another for that less acidic ammonium function (pKa2 = 8. 25.2: Isoelectric Points and ElectrophoresisĪfter completing this section, you should be able to.Īs noted earlier, the titration curves of easy proteins display two inflection points, one because of the strongly acidic carboxyl group (pKa1 = 1. The net charge on the molecule is affected by pH of its surrounding environment and can become more positively or negatively charged due to the gain or loss, respectively, of protons (H+). 
The standard nomenclature to represent the isoelectric point is pH(I). The isoelectric point (pI, pH(I), IEP), is the pH at which a molecule carries no net electrical charge or is electrically neutral in the statistical mean. You can practice this approach by designing your own polypeptides and check you answer using Thermo-Fisher’s peptide analyzer at This video walks you through my strategy for determining the isoelectic point (or pI) of a small polypeptide, and applies this strategy to an example. Video advice: Determining the isoelectric point of a polypeptide Moreover, the precise measurement of isoelectric points can be difficult, thus many sources often cite differing values for isoelectric points of these materials. The exact value can vary widely, depending on material factors such as purity and phase as well as physical parameters such as temperature. Note: The following list gives the isoelectric point at 25 ☌ for selected materials in water. 25.2: Isoelectric Points and Electrophoresis.IF there are charged side chains it is between the side chain of the first amino acid and the NH group of the second amino acid, unless the side groups are arginine, lysine, and tyrosine, then those side group pka are above the NH pka. General Rule I find is that between 2 separate amino acids that share a peptide bond, with out a charged side group, the pI is between the carboxylic acid of amino acid 1 and the pka of the NH group of amino acid 2. 
Then you add base after the second pka2 and see how the charges are if they are all negative the pi is not in this region. Then write a new column of adding base between pka1 and pka2 then see what happens to the charges there IF the charges equal 0 then your pi is around this mark in which case you do the following Then you write a new column with base added, 0 to first pka and then see what happens to charges on the strand, count them and see if they equal 0 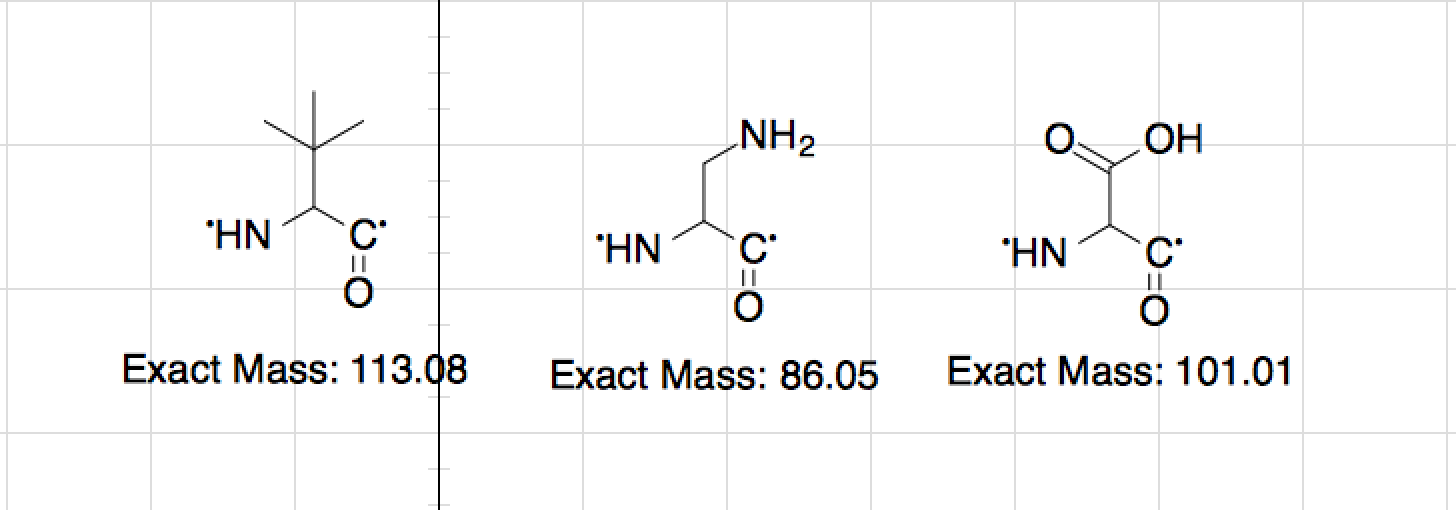
Write your strand in this case you chose I and N, so write out the structure and put the pka right next to each structure then start this as 0 base added If you are trying to find the PI this is what I would do
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
